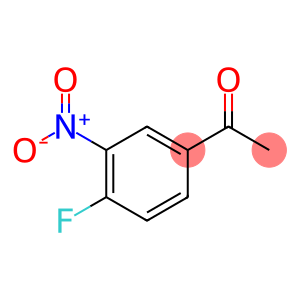
Ethanone, 1-(4-fluoro-3-nitrophenyl)-
4'-Fluoro-3'-nitroacetophenone
CAS: 400-93-1
Molecular Formula: C8H6FNO3
Ethanone, 1-(4-fluoro-3-nitrophenyl)- - Names and Identifiers
| Name | 4'-Fluoro-3'-nitroacetophenone |
| Synonyms | 4-Fluoro-3-nitroacetophneone 4-Fluoro-3-nitroacetophenone 3-nitro-4-fluoroacetophenone 4′-Fluor-3′-nitroacetophenon 4'-Fluoro-3'-notroacetophenone 4'-FLUORO-3'-NITROACETOPHENONE 4'-Fluoro-3'-nitroacetophenone 3'-nitro-4'-fluoroacetophenone 1-(4-fluoro-3-nitrophenyl) ethanone Ethanone, 1-(4-fluoro-3-nitrophenyl)- 1-(4-Fluoro-3-nitrophenyl)ethan-1-one 1-(4-Fluoro-3-nitrophenyl)ethan-1-one, 5-Acetyl-2-fluoronitrobenzene |
| CAS | 400-93-1 |
| EINECS | 626-780-8 |
| InChI | InChI=1/C7H5F3N2O2/c8-7(9,10)4-1-2-5(11)6(3-4)12(13)14/h1-3H,11H2 |
| InChIKey | PTCNZDJJIOLIKQ-UHFFFAOYSA-N |
Ethanone, 1-(4-fluoro-3-nitrophenyl)- - Physico-chemical Properties
| Molecular Formula | C8H6FNO3 |
| Molar Mass | 183.14 |
| Density | 1.336±0.06 g/cm3(Predicted) |
| Melting Point | 47-51 °C |
| Boling Point | 118-120°C 0,8mm |
| Flash Point | 118-120°C/0.8mm |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.00908mmHg at 25°C |
| Appearance | Bright yellow crystalline powder |
| Color | Light yellow |
| BRN | 2211976 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.525 |
| MDL | MFCD00115369 |
Ethanone, 1-(4-fluoro-3-nitrophenyl)- - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| HS Code | 29143990 |
| Hazard Class | IRRITANT |
Ethanone, 1-(4-fluoro-3-nitrophenyl)- - Reference Information
| use | 4 '-fluoro-3'-nitroacetophenone is an important organic fluorine-containing intermediate, which is mainly used to develop new fluorine-containing antibiotics, fluorobenzene salicylic acid, insecticides and liquid crystal materials, etc. The market demand is increasing year by year. |
| preparation | mix 50g magnesium nitrate, 40g ferric nitrate and 25g antimony nitrate, dissolve with 750mL deionized water to form a mixed salt solution, add 100gAl2O3 powder, and stir evenly; Dissolve 65g of sodium carbonate with 300mL of deionized water into a mixed alkali solution, and control the heating temperature between 60 ℃ and 65 ℃, drop into the mixed salt solution under stirring, control pH = 6.5, control the addition time for 4 hours, and generate precipitation at the same time. The precipitation is filtered and washed until it does not contain Na ions, extruded and formed, dried overnight in a drying oven at 110 ℃, and calcined at 450±5 ℃ for 2 hours to obtain the required catalyst I. The contents of magnesium oxide, iron oxide and antimony oxide in the catalyst I are 8.2%, 16.2% and 14.5% respectively. 4 '-fluoro -3'-nitroacetophenone is prepared by oxidative rearrangement hydrolysis of 4-fluoro -3-nitrophenol using the prepared catalyst I, 15 g4-fluoro -3-nitrophenol, 40mL acetonitrile and 2.5g catalyst I are fully mixed and added to the reactor, the temperature is raised to 50 ℃, 40mL 35% H2O2 is added dropwise, and the reaction is stirred for 10 hours at this temperature, the reaction solution is cooled and centrifuged, the catalyst is filtered out, 100mL of water is added to the filtrate for dilution, ether extraction, and the organic phase is dried with anhydrous magnesium sulfate and then distilled under reduced pressure to obtain a solid product 4 '-fluoro-3'-nitroacetophenone. The yield is 81.8%. |
Last Update:2024-04-09 15:16:50
Supplier List
Spot supply
Product Name: 4'-Fluoro-3'-nitroacetophenone Visit Supplier Webpage Request for quotationCAS: 400-93-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4'-Fluoro-3'-nitroacetophenone Request for quotation
CAS: 400-93-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 400-93-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4'-Fluoro-3'-nitroacetophenone Request for quotation
CAS: 400-93-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 400-93-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 4'-Fluoro-3'-nitroacetophenone Visit Supplier Webpage Request for quotation
CAS: 400-93-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 400-93-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4'-Fluoro-3'-nitroacetophenone Visit Supplier Webpage Request for quotationCAS: 400-93-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4'-Fluoro-3'-nitroacetophenone Request for quotation
CAS: 400-93-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 400-93-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4'-Fluoro-3'-nitroacetophenone Request for quotation
CAS: 400-93-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 400-93-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 4'-Fluoro-3'-nitroacetophenone Visit Supplier Webpage Request for quotation
CAS: 400-93-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 400-93-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



